Generally, de novo synthesis of fatty acids is strongly upregulated in tumor cells, due to the upregulation of the enzymes acetyl-CoA carboxylase and fatty acid synthetase.
Furthermore, the enzymes involved in the citrate shuttle and NADPH production, such as citrate lyase, NADP-dependent isocitrate dehydrogenase and NADP-dependent malic enzyme, are strongly upregulated in tumor cells.
To prevent an accumulation of fatty acids, special alterations take place during tumor formation.
Tumor pre-stages in the liver are able to accumulate triglycerides in large amounts, whereas later tumor stages cannot.
One metabolic cause for this alteration is the loss of cytosolic glycerol 3-P dehydrogenase.
Glycerol 3-P dehydrogenase is part of the glycerol 3-P shuttle and catalyzes the synthesis of glycerol 3-P from dihydroxyacetone-P (DHAP).
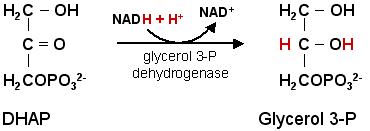
Glycerol 3-P is the backbone of triglycerides.
Dihydroxyacetone-P is the backbone of ether-linked glycerolipids.
In tumor cells, the strong reduction of glycerol 3-P dehydrogenase leads to an increase in fatty acid release.
The accumulation of dihydroxyacetone-P leads to an increased
ether-linked glycerolipid synthesis.
The release of fatty acids and the accumulation of ether-linked lipids may protect tumor cells from immune attack.